Astrocyte
| Astrocyte | |
|---|---|
 An astrocytic cell from rat brain grown in tissue culture and stained with antibodies to GFAP (red) and vimentin (green). Both proteins are present in large amounts in the intermediate filaments of this cell, so the cell appears yellow. The blue material shows DNA visualized with DAPI stain, and reveals the nuclei of the astrocyte and other cells. Image courtesy of EnCor Biotechnology Inc. | |
| Details | |
| Location | Brain and Spinal cord |
| Identifiers | |
| Latin | Astrocytus |
| MeSH | D001253 |
NeuroLex ID | sao1394521419 |
| TH | H2.00.06.2.00002, H2.00.06.2.01008 |
Anatomical terms of microanatomy [edit on Wikidata] | |
Astrocytes (Astro from Greek astron = star and cyte from Greek "kytos" = cavity but also means cell), also known collectively as astroglia, are characteristic star-shaped glial cells in the brain and spinal cord. The proportion of astrocytes in the brain is not well defined. Depending on the counting technique used, studies have found that the astrocyte proportion varies by region and ranges from 20% to 40% of all glia.[1] They perform many functions, including biochemical support of endothelial cells that form the blood–brain barrier, provision of nutrients to the nervous tissue, maintenance of extracellular ion balance, and a role in the repair and scarring process of the brain and spinal cord following traumatic injuries.
Research since the mid-1990s has shown that astrocytes propagate intercellular Ca2+ waves over long distances in response to stimulation, and, similar to neurons, release transmitters (called gliotransmitters) in a Ca2+-dependent manner.[2] Data suggest that astrocytes also signal to neurons through Ca2+-dependent release of glutamate.[3] Such discoveries have made astrocytes an important area of research within the field of neuroscience.
Contents
1 Structure
1.1 Development
2 Function
2.1 Tripartite synapse
3 Clinical significance
3.1 Astrocytomas
3.2 Neurodevelopment disorders
3.3 Chronic pain
3.3.1 Two states of persistent pain
3.4 Other pathologies
4 Research
5 Classification
5.1 Lineage and antigenic phenotype
5.2 Anatomical classification
5.3 Transporter/receptor classification
6 See also
7 References
8 Further reading
9 External links
Structure

Astrocytes (green) in the context of neurons (red) in a mouse cortex cell culture

23-week-old fetal brain culture human astrocyte
Astrocytes (red-yellow) among neurons (green) in the living cerebral cortex
Astrocytes are a sub-type of glial cells in the central nervous system. They are also known as astrocytic glial cells. Star-shaped, their many processes envelop synapses made by neurons. Astrocytes are classically identified using histological analysis; many of these cells express the intermediate filament glial fibrillary acidic protein (GFAP).[4] Several forms of astrocytes exist in the central nervous system including fibrous (in white matter), protoplasmic (in grey matter), and radial. The fibrous glia are usually located within white matter, have relatively few organelles, and exhibit long unbranched cellular processes. This type often has "vascular feet" that physically connect the cells to the outside of capillary walls when they are in proximity to them. The protoplasmic glia are the most prevalent and are found in grey matter tissue, possess a larger quantity of organelles, and exhibit short and highly branched tertiary processes. The radial glia are disposed in planes perpendicular to the axes of ventricles. One of their processes abuts the pia mater, while the other is deeply buried in gray matter. Radial glia are mostly present during development, playing a role in neuron migration. Müller cells of the retina and Bergmann glia cells of the cerebellar cortex represent an exception, being present still during adulthood. When in proximity to the pia mater, all three forms of astrocytes send out processes to form the pia-glial membrane.
Development

Astrocytes are depicted in red. Cell nuclei are depicted in blue. Astrocytes were obtained from brains of newborn mice
Astrocytes are macroglial cells in the central nervous system. Astrocytes are derived from heterogeneous populations of progenitor cells in the neuroepithelium of the developing central nervous system. There is remarkable similarity between the well known genetic mechanisms that specify the lineage of diverse neuron subtypes and that of macroglial cells.[5] Just as with neuronal cell specification, canonical signaling factors like Sonic hedgehog (SHH), Fibroblast growth factor (FGFs), WNTs and bone morphogenetic proteins (BMPs), provide positional information to developing macroglial cells through morphogen gradients along the dorsal–ventral, anterior–posterior and medial–lateral axes. The resultant patterning along the neuraxis leads to segmentation of the neuroepithelium into progenitor domains (p0, p1 p2, p3 and pMN) for distinct neuron types in the developing spinal cord. On the basis of several studies it is now believed that this model also applies to macroglial cell specification. Studies carried out by Hochstim and colleagues have demonstrated that three distinct populations of astrocytes arise from the p1, p2 and p3 domains.[6] These subtypes of astrocytes can be identified on the basis of their expression of different transcription factors (PAX6, NKX6.1) and cell surface markers (reelin and SLIT1). The three populations of astrocyte subtypes which have been identified are 1) dorsally located VA1 astrocytes, derived from p1 domain, express PAX6 and reelin 2) ventrally located VA3 astrocytes, derived from p3, express NKX6.1 and SLIT1 and 3) and intermediate white-matter located VA2 astrocyte, derived from the p2 domain, which express PAX6, NKX6.1, reelin and SLIT1.[7] After astrocyte specification has occurred in the developing CNS, it is believed that astrocyte precursors migrate to their final positions within the nervous system before the process of terminal differentiation occurs.
Function
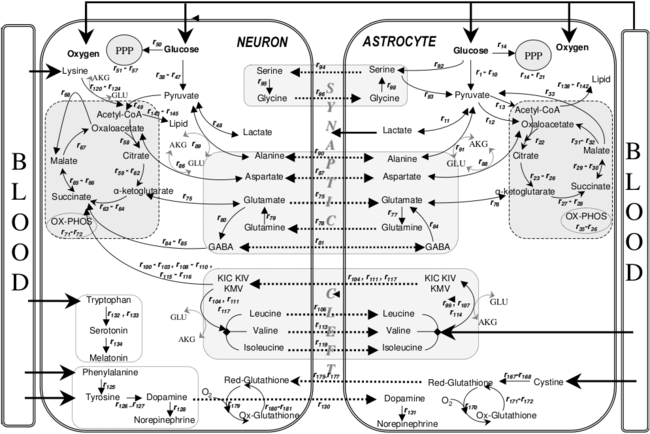
Metabolic interactions between astrocytes and neurons.[8]
Previously in medical science, the neuronal network was considered the only important function of astrocytes, and they were looked upon as gap fillers. More recently, the function of astrocytes has been reconsidered, and they are now thought to play a number of active roles in the brain, including the secretion or absorption of neural transmitters and maintenance of the blood–brain barrier.[9] Following on this idea the concept of a tripartite synapse has been proposed, referring to the tight relationship occurring at synapses among a presynaptic element, a postsynaptic element and a glial element.[10]
Structural: They are involved in the physical structuring of the brain. Astrocytes get their name because they are "star-shaped". They are the most abundant glial cells in the brain that are closely associated with neuronal synapses. They regulate the transmission of electrical impulses within the brain.
Glycogen fuel reserve buffer: Astrocytes contain glycogen and are capable of gluconeogenesis. The astrocytes next to neurons in the frontal cortex and hippocampus store and release glucose. Thus, astrocytes can fuel neurons with glucose during periods of high rate of glucose consumption and glucose shortage. A recent research on rats suggests there may be a connection between this activity and physical exercise.[11]
Metabolic support: They provide neurons with nutrients such as lactate.
Glucose sensing: normally associated with neurons, the detection of interstitial glucose levels within the brain is also controlled by astrocytes. Astrocytes in vitro become activated by low glucose and are in vivo this activation increases gastric emptying to increase digestion.[12]
Blood–brain barrier: The astrocyte end-feet encircling endothelial cells were thought to aid in the maintenance of the blood–brain barrier, but recent research indicates that they do not play a substantial role; instead, it is the tight junctions and basal lamina of the cerebral endothelial cells that play the most substantial role in maintaining the barrier.[13] However, it has recently been shown that astrocyte activity is linked to blood flow in the brain, and that this is what is actually being measured in fMRI.[14][15]
Transmitter uptake and release: Astrocytes express plasma membrane transporters such as glutamate transporters for several neurotransmitters, including glutamate, ATP, and GABA. More recently, astrocytes were shown to release glutamate or ATP in a vesicular, Ca2+-dependent manner.[16] (This has been disputed for hippocampal astrocytes.)[17]
Regulation of ion concentration in the extracellular space: Astrocytes express potassium channels at a high density. When neurons are active, they release potassium, increasing the local extracellular concentration. Because astrocytes are highly permeable to potassium, they rapidly clear the excess accumulation in the extracellular space.[18] If this function is interfered with, the extracellular concentration of potassium will rise, leading to neuronal depolarization by the Goldman equation. Abnormal accumulation of extracellular potassium is well known to result in epileptic neuronal activity.[19]
Modulation of synaptic transmission: In the supraoptic nucleus of the hypothalamus, rapid changes in astrocyte morphology have been shown to affect heterosynaptic transmission between neurons.[20] In the hippocampus, astrocytes suppress synaptic transmission by releasing ATP, which is hydrolyzed by ectonucleotidases to yield adenosine. Adenosine acts on neuronal adenosine receptors to inhibit synaptic transmission, thereby increasing the dynamic range available for LTP.[21]
Vasomodulation: Astrocytes may serve as intermediaries in neuronal regulation of blood flow.[22]
Promotion of the myelinating activity of oligodendrocytes: Electrical activity in neurons causes them to release ATP, which serves as an important stimulus for myelin to form. However, the ATP does not act directly on oligodendrocytes. Instead, it causes astrocytes to secrete cytokine leukemia inhibitory factor (LIF), a regulatory protein that promotes the myelinating activity of oligodendrocytes. This suggests that astrocytes have an executive-coordinating role in the brain.[23]
Nervous system repair: Upon injury to nerve cells within the central nervous system, astrocytes fill up the space to form a glial scar, and may contribute to neural repair. The role of astrocytes in CNS regeneration following injury is not well understood though. The glial scar has traditionally been described as an impermeable barrier to regeneration, thus implicating a negative role in axon regeneration. However, recently, it was found through genetic ablation studies that astrocytes are actually required for regeneration to occur.[24] More importantly, the authors found that the astrocyte scar is actually essential for stimulated axons (that axons that have been coaxed to grow via neurotrophic supplementation) to extend through the injured spinal cord.[24] Astrocytes that have been pushed into a reactive phenotype (termed astrogliosis, defined by upregulation of GFAP expression, a definition still under debate) may actually be toxic to neurons, releasing signals that can kill neurons.[25] Much work, however, remains to elucidate their role in nervous system injury.
Long-term potentiation: Scientists debate whether astrocytes integrate learning and memory in the hippocampus. Recently it has been shown that engrafting human glial progenitor cell in the nascent mice brains will cause the cells to differentiate into astrocytes. After differentiation these cells increase LTP and improve memory performance in the mice.[26]
The switch of the nervous system: Based on the evidence listed below, it has been recently conjectured in,[27] that macro glia (and astrocytes in particular) act both as a lossy neurotransmitter capacitor and as the logical switch of the nervous system. I.e., macroglia either block or enable the propagation of the stimulus along the nervous system, depending on their membrane state and the level of the stimulus.

Fig. 6 The conjectured switching role of glia in the biological neural detection scheme as suggested by Nossenson et al[27][28]
| Evidence type | Description | References |
|---|---|---|
| Calcium evidence | Calcium waves appear only if a certain concentration of neurotransmitter is exceeded | [29][30][31] |
| Electrophysiological evidence | A negative wave appears when the stimulus level crosses a certain threshold. The shape of the electrophysiological response is different and has the opposite polarity compared to the characteristic neural response, suggesting that cells other than neurons might be involved. | [32][33] [34][35] |
| Psychophysical evidence | The negative electrophysiological response is accompanied with all-or-none actions. A moderate negative electrophysiological response appears in conscious logical decisions such as perception tasks. An intense sharp negative wave appear in epileptic seizures and during reflexes. | [32][35][33][34] |
| Radioactivity based glutamate uptake tests | Glutamate uptake tests indicate that astrocyte process glutamate in a rate which is initially proportional to glutamate concentration. This supports the leaky capacitor model, where the 'leak' is glutamate processing by glia's glutamine synthetase. Furthermore, the same tests indicate on a saturation level after which neurotransmitter uptake level stops rising proportionally to neurotransmitter concentration. The latter supports the existence of a threshold. The graphs which show these characteristics are referred to as Michaelis-Menten graphs | [36] |
Astrocytes are linked by gap junctions, creating an electrically coupled (functional) syncytium.[37] Because of this ability of astrocytes to communicate with their neighbors, changes in the activity of one astrocyte can have repercussions on the activities of others that are quite distant from the original astrocyte.
An influx of Ca2+ ions into astrocytes is the essential change that ultimately generates calcium waves. Because this influx is directly caused by an increase in blood flow to the brain, calcium waves are said to be a kind of hemodynamic response function. An increase in intracellular calcium concentration can propagate outwards through this functional syncytium. Mechanisms of calcium wave propagation include diffusion of calcium ions and IP3 through gap junctions and extracellular ATP signalling.[38] Calcium elevations are the primary known axis of activation in astrocytes, and are necessary and sufficient for some types of astrocytic glutamate release.[39] Given the importance of calcium signaling in astrocytes, tight regulatory mechanisms for the progression of the spatio-temporal calcium signaling have been developed. Via mathematical analysis it has been shown that localized inflow of Ca2+ ions yields a localized raise in the cytosolic concentration of Ca2+ ions.[40] Moreover, cytosolic Ca2+ accumulation is independent of every intracellular calcium flux and depends on the Ca2+ exchange across the
membrane, cytosolic calcium diffusion, geometry of the cell, extracellular calcium perturbation, and initial concentrations.[40]
Tripartite synapse
Within the dorsal horn of the spinal cord, activated astrocytes have the ability to respond to almost all neurotransmitters[41] and, upon activation, release a multitude of neuroactive molecules such as glutamate, ATP, nitric oxide (NO), and prostaglandins (PG), which in turn influences neuronal excitability. The close association between astrocytes and presynaptic and postsynaptic terminals as well as their ability to integrate synaptic activity and release neuromodulators has been termed the tripartite synapse.[10] Synaptic modulation by astrocytes takes place because of this three-part association.
Clinical significance
Astrocytomas
Astrocytomas are primary intracranial tumors derived from astrocytes cells of the brain. It is also possible that glial progenitors or neural stem cells give rise to astrocytomas.
Astrocytomas are brain tumors that develop from astrocytes. They may occur in many parts of the brain and sometimes in the spinal cord. They can occur at any age and they primarily occur in males. Astrocytomas are divided into two categories: Low grade (I and II) and High Grade (III and IV). Low grade tumors are more common in children and high grade tumors are more common in adults.[42]
Pilocytic astrocytoma are Grade I tumors. They are considered benign and slow growing tumors. Pilocytic astrocytomas frequently have cystic portions filled with fluid and a nodule, which is the solid portion. Most are located in the cerebellum. Therefore, most symptoms are related to balance or coordination difficulties.[42] They also occur more frequently in children and teens.[43]
Grade II Tumors grow relatively slow but invade surrounding healthy tissue. Usually considered benign but can grow into malignant tumors. Other names that are used are Fibrillary or Protoplasmic astrocytomas. They are prevalent in younger people who are often present with seizures.[43]
Anaplastic astrocytoma is classified as grade III and are malignant tumors. They grow more rapidly than lower grade tumors and tend to invade nearby healthy tissue. Anaplastic astrocytomas recur more frequently than lower grade tumors because their tendency to spread into surrounding tissue makes them difficult to completely remove surgically.[42]
Glioblastoma Multiforme is also a malignant tumor and classified as a grade IV. Glioblastomas can contain more than one cell type (i.e., astrocytes, oligodendrocytes). Also, while one cell type may die off in response to a particular treatment, the other cell types may continue to multiply. Glioblastomas are the most invasive type of glial tumors as they grow rapidly and spread to nearby tissue. Approximately 50% of astrocytomas are glioblastomas and are very difficult to treat.[42]
Neurodevelopment disorders
A relatively new direction has emerged in the field of neurodevelopmental disorders. This view states that astrocyte dysfunction may play a role in the aberration of neuronal circuitry that underlies certain psychiatric disorders, including the autism spectrum disorders (ASDs) and schizophrenia.[44][45]
Chronic pain
Under normal conditions, pain conduction begins with some noxious signal followed by an action potential carried by nociceptive (pain sensing) afferent neurons, which elicit excitatory postsynaptic potentials (EPSP) in the dorsal horn of the spinal cord. That message is then relayed to the cerebral cortex, where we translate those EPSPs into "pain." Since the discovery of astrocytic influence, our understanding of the conduction of pain has been dramatically complicated. Pain processing is no longer seen as a repetitive relay of signals from body to brain, but as a complex system that can be up- and down-regulated by a number of different factors. One factor at the forefront of recent research is in the pain-potentiating synapse located in the dorsal horn of the spinal cord and the role of astrocytes in encapsulating these synapses. Garrison and co-workers[46] were the first to suggest association when they found a correlation between astrocyte hypertrophy in the dorsal horn of the spinal cord and hypersensitivity to pain after peripheral nerve injury, typically considered an indicator of glial activation after injury. Astrocytes detect neuronal activity and can release chemical transmitters, which in turn control synaptic activity.[41][47][48] In the past, hyperalgesia was thought to be modulated by the release of substance P and excitatory amino acids (EAA), such as glutamate, from the presynaptic afferent nerve terminals in the spinal cord dorsal horn. Subsequent activation of AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid), NMDA (N-methyl-D-aspartate) and kainate subtypes of ionotropic glutamate receptors follows. It is the activation of these receptors that potentiates the pain signal up the spinal cord. This idea, although true, is an oversimplification of pain transduction. A litany of other neurotransmitter and neuromodulators, such as calcitonin gene-related peptide (CGRP), adenosine triphosphate (ATP), brain-derived neurotrophic factor (BDNF), somatostatin, vasoactive intestinal peptide (VIP), galanin, and vasopressin are all synthesized and released in response to noxious stimuli. In addition to each of these regulatory factors, several other interactions between pain-transmitting neurons and other neurons in the dorsal horn have added impact on pain pathways.
Two states of persistent pain
After persistent peripheral tissue damage there is a release of several factors from the injured tissue as well as in the spinal dorsal horn. These factors increase the responsiveness of the dorsal horn pain-projection neurons to ensuing stimuli, termed "spinal sensitization," thus amplifying the pain impulse to the brain. Release of glutamate, substance P, and calcitonin gene-related peptide (CGRP) mediates NMDAR activation (originally silent because it is plugged by Mg2+), thus aiding in depolarization of the postsynaptic pain-transmitting neurons (PTN). In addition, activation of IP3 signaling and MAPKs (mitogen-activated protein kinases) such as ERK and JNK, bring about an increase in the synthesis of inflammatory factors that alter glutamate transporter function. ERK also further activates AMPARs and NMDARs in neurons. Nociception is further sensitized by the association of ATP and substance P with their respective receptors (P2X3) and neurokinin 1 receptor (NK1R), as well as activation of metabotropic glutamate receptors and release of BDNF. Persistent presence of glutamate in the synapse eventually results in dysregulation of GLT1 and GLAST, crucial transporters of glutamate into astrocytes. Ongoing excitation can also induce ERK and JNK activation, resulting in release of several inflammatory factors.
As noxious pain is sustained, spinal sensitization creates transcriptional changes in the neurons of the dorsal horn that lead to altered function for extended periods. Mobilization of Ca2+ from internal stores results from persistent synaptic activity and leads to the release of glutamate, ATP, tumor necrosis factor-α (TNF-α), interleukin 1β (IL-1β), IL-6, nitric oxide (NO), and prostaglandin E2 (PGE2). Activated astrocytes are also a source of matrix metalloproteinase 2 (MMP2), which induces pro-IL-1β cleavage and sustains astrocyte activation. In this chronic signaling pathway, p38 is activated as a result of IL-1β signaling, and there is a presence of chemokines that trigger their receptors to become active. In response to nerve damage, heat shock proteins (HSP) are released and can bind to their respective TLRs, leading to further activation.
Other pathologies
Other clinically significant pathologies involving astrocytes include astrogliosis and astrocytopathy.[49]
Research
A study performed in November 2010 and published March 2011, was done by a team of scientists from the University of Rochester and University of Colorado School of Medicine. They did an experiment to attempt to repair trauma to the Central Nervous System of an adult rat by replacing the glial cells. When the glial cells were injected into the injury of the adult rat’s spinal cord, astrocytes were generated by exposing human glial precursor cells to bone morphogenetic protein (Bone morphogenetic protein is important because it is considered to create tissue architecture throughout the body). So, with the bone protein and human glial cells combined, they promoted significant recovery of conscious foot placement, axonal growth, and obvious increases in neuronal survival in the spinal cord laminae. On the other hand, human glial precursor cells and astrocytes generated from these cells by being in contact with ciliary neurotrophic factors, failed to promote neuronal survival and support of axonal growth at the spot of the injury.[50]
One study done in Shanghai had two types of hippocampal neuronal cultures: In one culture, the neuron was grown from a layer of astrocytes and the other culture was not in contact with any astrocytes, but they were instead fed a glial conditioned medium (GCM), which inhibits the rapid growth of cultured astrocytes in the brains of rats in most cases. In their results they were able to see that astrocytes had a direct role in Long-term potentiation with the mixed culture (which is the culture that was grown from a layer of astrocytes) but not in GCM cultures.[51]
Studies have shown that astrocytes play an important function in the regulation of neural stem cells. Research from the Schepens Eye Research Institute at Harvard shows the human brain to abound in neural stem cells, which are kept in a dormant state by chemical signals (ephrin-A2 and ephrin-A3) from the astrocytes. The astrocytes are able to activate the stem cells to transform into working neurons by dampening the release of ephrin-A2 and ephrin-A3.[52]
In a study published in a 2011 issue of Nature Biotechnology[53] a group of researchers from the University of Wisconsin reports that it has been able to direct embryonic and induced human stem cells to become astrocytes.
A 2012 study[54] of the effects of marijuana on short term memories found that THC activates CB1 receptors of astrocytes which cause receptors for AMPA to be removed from the membranes of associated neurons.
Classification
There are several different ways to classify astrocytes.
Lineage and antigenic phenotype
These have been established by classic work by Raff et al. in early 1980s on Rat optic nerves.
- Type 1: Antigenically Ran2+, GFAP+, FGFR3+, A2B5−, thus resembling the "type 1 astrocyte" of the postnatal day 7 rat optic nerve. These can arise from the tripotential glial restricted precursor cells (GRP), but not from the bipotential O2A/OPC (oligodendrocyte, type 2 astrocyte precursor, also called Oligodendrocyte progenitor cell) cells.
- Type 2: Antigenically A2B5+, GFAP+, FGFR3−, Ran 2−. These cells can develop in vitro from the either tripotential GRP (probably via O2A stage) or from bipotential O2A cells (which some people{{[55]}} think may in turn have been derived from the GRP) or in vivo when these progenitor cells are transplanted into lesion sites (but probably not in normal development, at least not in the rat optic nerve). Type-2 astrocytes are the major astrocytic component in postnatal optic nerve cultures that are generated by O2A cells grown in the presence of fetal calf serum but are not thought to exist in vivo.[56]
Anatomical classification
- Protoplasmic: found in grey matter and have many branching processes whose end-feet envelop synapses. Some protoplasmic astrocytes are generated by multipotent subventricular zone progenitor cells.[57][58]
- Gömöri-positive astrocytes. These are a subset of protoplasmic astrocytes that contain numerous cytoplasmic inclusions, or granules, that stain positively with Gömöri's chrome-alum hematoxylin stain. It is now known that these granules are formed from the remnants of degenerating mitochondria engulfed within lysosomes,[59] Some type of oxidative stress appears to be responsible for the mitochondrial damage within these specialized astrocytes. Gömöri-positive astrocytes are much more abundant within the arcuate nucleus of the hypothalamus and in the hippocampus than in other brain regions. They may have a role in regulating the response of the hypothalamus to glucose.[60][61]
- Fibrous: found in white matter and have long thin unbranched processes whose end-feet envelop nodes of Ranvier. Some fibrous astrocytes are generated by radial glia.[62][63][64][65][66]
Transporter/receptor classification
- GluT type: these express glutamate transporters (EAAT1/SLC1A3 and EAAT2/SLC1A2) and respond to synaptic release of glutamate by transporter currents. The function and availability of EAAT2 is modulated by TAAR1, an intracellular receptor in human astrocytes.[67]
- GluR type: these express glutamate receptors (mostly mGluR and AMPA type) and respond to synaptic release of glutamate by channel-mediated currents and IP3-dependent Ca2+ transients.
See also
- Bergmann gliosis
- Radial glial cell
- Neuroglia
- Pituicyte
- Polydendrocytes
- List of human cell types derived from the germ layers
References
^ Verkhratsky, A.; Butt, A.M. (2013). "Numbers: how many glial cells are in the brain?". Glial Physiology and Pathophysiology. John Wiley and Sons. pp. 93–96. ISBN 978-0-470-97853-5..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ "Role of Astrocytes in the Central Nervous System". Retrieved 27 July 2018.
^ Fiacco TA, Agulhon C, McCarthy KD (October 2008). "Sorting out Astrocyte Physiology from Pharmacology". Annu. Rev. Pharmacol. Toxicol. 49 (1): 151–74. doi:10.1146/annurev.pharmtox.011008.145602. PMID 18834310.
^ Sarma, PotukuchiVenkata Gurunadha Krishna; Srikanth, Lokanathan; Vengamma, Bhuma; Venkatesh, Katari; Chandrasekhar, Chodimella; Mouleshwara Prasad, BodapatiChandra; Sanjeevkumar, Akula (2013). "In vitro differentiation of cultured human CD34+ cells into astrocytes". Neurology India. 61 (4): 383–8. doi:10.4103/0028-3886.117615. PMID 24005729.
^ Rowitch, David H.; Kriegstein, Arnold R. (2010). "Developmental genetics of vertebrate glial–cell specification". Nature. 468 (7321): 214–22. Bibcode:2010Natur.468..214R. doi:10.1038/nature09611. PMID 21068830.
^ Muroyama, Y; Fujiwara, Y; Orkin, SH; Rowitch, DH (2005). "Specification of astrocytes by bHLH protein SCL in a restricted region of the neural tube". Nature. 438 (7066): 360–363. Bibcode:2005Natur.438..360M. doi:10.1038/nature04139. PMID 16292311.
^ Hochstim, C; Deneen, B; Lukaszewicz, A; Zhou, Q; Anderson, DJ (2008). "Identification of positionally distinct astrocyte subtypes whose identities are specified by a homeodomain code". Cell. 133 (3): 510–522. doi:10.1016/j.cell.2008.02.046. PMC 2394859. PMID 18455991.
^ Çakιr, Tunahan; Alsan, Selma; Saybaşιlι, Hale; Akιn, Ata; Ülgen, Kutlu Ö (2007). "Reconstruction and flux analysis of coupling between metabolic pathways of astrocytes and neurons: application to cerebral hypoxia". Theoretical Biology and Medical Modelling. 4 (1): 48. doi:10.1186/1742-4682-4-48. PMC 2246127. PMID 18070347.
^ Kolb, Brian and Whishaw, Ian Q. (2008) Fundamentals of Human Neuropsychology. Worth Publishers. 6th ed.
ISBN 0716795868
^ ab Araque A, Parpura V, Sanzgiri RP, Haydon PG (1999). "Tripartite synapses: glia, the unacknowledged partner". Trends in Neurosciences. 22 (5): 208–215. doi:10.1016/S0166-2236(98)01349-6. PMID 10322493.
^ Reynolds, Gretchen (22 February 2012). "How Exercise Fuels the Brain". New York Times.
^ McDougal, David H.; Viard, Edouard; Hermann, Gerlinda E.; Rogers, Richard C. (April 2013). "Astrocytes in the hindbrain detect glucoprivation and regulate gastric motility". Autonomic neuroscience : basic & clinical. 175 (0): 61–69. doi:10.1016/j.autneu.2012.12.006. ISSN 1566-0702. PMC 3951246. PMID 23313342.
^ Kimelberg HK, Jalonen T, Walz W (1993). "Regulation of the brain microenvironment:transmitters and ions.". In Murphy S. Astrocytes: pharmacology and function. San Diego, CA: Academic Press. pp. 193–222. ISBN 978-0125113700.
^ Swaminathan N (1 October 2008). "Brain-scan mystery solved". Scientific American Mind: 7.
^ Figley CR, Stroman PW (February 2011). "The role(s) of astrocytes and astrocyte activity in neurometabolism, neurovascular coupling, and the production of functional neuroimaging signals". The European Journal of Neuroscience. 33 (4): 577–88. doi:10.1111/j.1460-9568.2010.07584.x. PMID 21314846.
^ Santello M, Volterra A (2008). "Synaptic modulation by astrocytes via Ca2+-dependent glutamate release". Neuroscience. Mar. 158 (1): 253–9. doi:10.1016/j.neuroscience.2008.03.039. PMID 18455880.
^ Agulhon C, Fiacco T, McCarthy K (2010). "Hippocampal short- and long-term plasticity are not modulated by astrocyte Ca2+ signaling". Science. 327 (5970): 1250–1257. Bibcode:2010Sci...327.1250A. doi:10.1126/science.1184821. PMID 20203048.
^ Walz W (2000). "Role of astrocytes in the clearance of excess extracellular potassium". Neurochemistry International. 36 (4–5): 291–300. doi:10.1016/S0197-0186(99)00137-0. PMID 10732996.
^ Gabriel S, Njunting M, Pomper JK, Merschhemke M, Sanabria ER, Eilers A, Kivi A, Zeller M, Meencke HJ, Cavalheiro EA, Heinemann U, Lehmann TN (2004). "Stimulus and Potassium-Induced Epileptiform Activity in the Human Dentate Gyrus from Patients with and without Hippocampal Sclerosis". The Journal of Neuroscience. 24 (46): 10416–10430. doi:10.1523/JNEUROSCI.2074-04.2004. PMID 15548657.
^ Piet R, Vargová L, Syková E, Poulain D, Oliet S (2004). "Physiological contribution of the astrocytic environment of neurons to intersynaptic crosstalk". Proceedings of the National Academy of Sciences of the United States of America. 101 (7): 2151–5. Bibcode:2004PNAS..101.2151P. doi:10.1073/pnas.0308408100. PMC 357067. PMID 14766975.
^ Pascual O, Casper KB, Kubera C, Zhang J, Revilla-Sanchez R, Sul JY, Takano H, Moss SJ, McCarthy K, Haydon PG (2005). "Astrocytic purinergic signaling coordinates synaptic networks". Science. 310 (5745): 113–6. Bibcode:2005Sci...310..113P. doi:10.1126/science.1116916. PMID 16210541.
^ Parri R, Crunelli V (2003). "An astrocyte bridge from synapse to blood flow". Nature Neuroscience. 6 (1): 5–6. doi:10.1038/nn0103-5. PMID 12494240.
^ Ishibashi T, Dakin K, Stevens B, Lee P, Kozlov S, Stewart C, Fields R (2006). "Astrocytes Promote Myelination in Response to Electrical Impulses". Neuron. 49 (6): 823–32. doi:10.1016/j.neuron.2006.02.006. PMC 1474838. PMID 16543131.
^ ab Anderson MA, Burda JE, Ren Y, Ao Y, O'Shea TM, Kawaguchi R, Coppola G, Khakh BS, Deming TJ, Sofroniew MV (April 2016). "Astrocyte scar formation aids central nervous system axon regeneration". Nature. 532 (7598): 195–200. Bibcode:2016Natur.532..195A. doi:10.1038/nature17623. PMC 5243141. PMID 27027288.
^ Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Münch AE, Chung WS, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM, Stevens B, Barres BA (January 2017). "Neurotoxic reactive astrocytes are induced by activated microglia". Nature. 541 (7638): 481–487. Bibcode:2017Natur.541..481L. doi:10.1038/nature21029. PMC 5404890. PMID 28099414.
^ Han, Xiaoning; Chen, Michael; Wang, Fushun; Windrem, Martha; Wang, Su; Shanz, Steven; Xu, Qiwu; Oberheim, Nancy Ann; Bekar, Lane (2013-07-03). "Forebrain Engraftment by Human Glial Progenitor Cells Enhances Synaptic Plasticity and Learning in Adult Mice". Cell Stem Cell. 12 (3): 342–353. doi:10.1016/j.stem.2012.12.015. ISSN 1934-5909. PMC 3700554. PMID 23472873.
^ abc Nossenson, N.; Magal, N. ,; Messer, H., (2016). "Detection of stimuli from multi-neuron activity: Empirical study and theoretical implications". Neurocomputing. 174: 822–837.
^ ab Nossenson, Nir (2013). Model Based Detection of a Stimulus Presence from Neurophysiological Signals (PDF). The Neiman Library of Exact Sciences & Engineering, Tel Aviv University: PhD diss, University of Tel-Aviv.
^ Cornell-Bell, AH; Finkbeiner, SM; Cooper, MS; Smith, SJ (26 January 1990). "Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling". Science. 247 (4941): 470–3. Bibcode:1990Sci...247..470C. doi:10.1126/science.1967852. PMID 1967852.
^ Jahromi, BS; Robitaille, R; Charlton, MP (June 1992). "Transmitter release increases intracellular calcium in perisynaptic Schwann cells in situ". Neuron. 8 (6): 1069–77. PMID 1351731.
^ Verkhratsky, A; Orkand, RK; Kettenmann, H (January 1998). "Glial calcium: homeostasis and signaling function". Physiological Reviews. 78 (1): 99–141. doi:10.1152/physrev.1998.78.1.99. PMID 9457170.
^ ab Ebert, U; Koch, M (September 1997). "Acoustic startle-evoked potentials in the rat amygdala: effect of kindling". Physiology & Behavior. 62 (3): 557–62. PMID 9272664.
^ ab Frot, M; Magnin, M; Mauguière, F; Garcia-Larrea, L (March 2007). "Human SII and posterior insula differently encode thermal laser stimuli". Cerebral Cortex. 17 (3): 610–20. doi:10.1093/cercor/bhk007. PMID 16614165.
^ ab Perlman, Ido. "The Electroretinogram: ERG by Ido Perlman – Webvision". webvision.med.utah.edu.
^ ab Tian, GF; Azmi, H; Takano, T; Xu, Q; Peng, W; Lin, J; Oberheim, N; Lou, N; Wang, X; Zielke, HR; Kang, J; Nedergaard, M (September 2005). "An astrocytic basis of epilepsy". Nature Medicine. 11 (9): 973–81. doi:10.1038/nm1277. PMC 1850946. PMID 16116433.
^ Hertz, L; Schousboe, A; Boechler, N; Mukerji, S; Fedoroff, S (February 1978). "Kinetic characteristics of the glutamate uptake into normal astrocytes in cultures". Neurochemical Research. 3 (1): 1–14. PMID 683409.
^ Bennett M, Contreras J, Bukauskas F, Sáez J (2003). "New roles for astrocytes: gap junction hemichannels have something to communicate". Trends Neurosci. 26 (11): 610–7. doi:10.1016/j.tins.2003.09.008. PMC 3694339. PMID 14585601.
^ Newman, E. A. (2001). "Propagation of intercellular calcium waves in retinal astrocytes and Müller cells". The Journal of Neuroscience. 21 (7): 2215–23. PMC 2409971. PMID 11264297.
^ Parpura V, Haydon P (2000). "Physiological astrocytic calcium levels stimulate glutamate release to modulate adjacent neurons". Proceedings of the National Academy of Sciences of the United States of America. 97 (15): 8629–34. Bibcode:2000PNAS...97.8629P. doi:10.1073/pnas.97.15.8629. PMC 26999. PMID 10900020.
^ ab Lopez-Caamal, F.; Oyarzun, D.A.; Middleton, R.H.; Garcia, M.R. (May 2014). "Spatial Quantification of Cytosolic Ca2+ Accumulation in Nonexcitable Cells:An Analytical Study". IEEE/ACM Transactions on Computational Biology and Bioinformatics. 11 (3): 592–603. doi:10.1109/TCBB.2014.2316010. PMID 26356026.
^ ab Haydon, Philip G. (2001). "Glia: listening and talking to the synapse" (PDF). Nature Reviews Neuroscience. 2 (3): 185–193. doi:10.1038/35058528. PMID 11256079.
^ abcd Astrocytomas. International RadioSurgery Association (2010).
^ ab Astrocytoma Tumors. American Association of Neurological Surgeons (August 2005).
^ Barker, AJ; Ullian, EM (2008). "New roles for astrocytes in developing synaptic circuits". Communicative & Integrative Biology. 1 (2): 207–11. doi:10.4161/cib.1.2.7284. PMC 2686024. PMID 19513261.
^ Sloan, SA; Barres, BA (Mar 29, 2014). "Mechanisms of astrocyte development and their contributions to neurodevelopmental disorders". Current Opinion in Neurobiology. 27C: 75–81. doi:10.1016/j.conb.2014.03.005. PMC 4433289. PMID 24694749.
^ Garrison, C.J.; Dougherty, P.M.; Kajander, K.C.; Carlton, S.M. (1991). "Staining of glial fibrillary acidic protein (GFAP) in lumbar spinal cord increases following a sciatic nerve constriction injury". Brain Research. 565 (1): 1–7. doi:10.1016/0006-8993(91)91729-K. PMID 1723019.
^ Volterra, A. & Meldolesi, J. (2005). "Astrocytes, from brain glue to communication elements: the revolution continues". Nature Reviews Neuroscience. 6 (8): 626–640. doi:10.1038/nrn1722. PMID 16025096.
^ Halassa, M.M.; Fellin, T. & Haydon, P.G. (2006). "The tripartite synapse: roles for gliotransmission in health and disease". Trends Mol. Med. 13 (2): 54–63. doi:10.1016/j.molmed.2006.12.005. PMID 17207662.
^ Sofroniew, Michael V (2015). "Astrogliosis". Cold Spring Harbor Perspectives in Biology. 7 (2): a020420. doi:10.1101/cshperspect.a020420. PMC 4315924. PMID 25380660.
^ Davies SJA; Shih C-H; Noble M; Mayer-Proschel M; Davies JE; et al. (2011). Combs, Colin, ed. "Transplantation of Specific Human Astrocytes Promotes Functional Recovery after spinal Cord Injury". PLoS ONE. 6 (3): e17328. Bibcode:2011PLoSO...617328D. doi:10.1371/journal.pone.0017328. PMC 3047562. PMID 21407803.
^ Yang, Y. (2003). "Contribution of astrocytes to hippocampal long-term potentiation through release of D-serine". Proceedings of the National Academy of Sciences of the United States of America. 100 (25): 15194–15199. Bibcode:2003PNAS..10015194Y. doi:10.1073/pnas.2431073100. PMC 299953. PMID 14638938.
^ Jiao JW, Feldheim DA, Chen DF (June 2008). "Ephrins as negative regulators of adult neurogenesis in diverse regions of the central nervous system". Proceedings of the National Academy of Sciences of the United States of America. 105 (25): 8778–83. Bibcode:2008PNAS..105.8778J. doi:10.1073/pnas.0708861105. PMC 2438395. PMID 18562299.
^ Krencik, Robert; Weick, Jason P; Liu, Yan; Zhang, Zhi-Jian; Zhang, Su-Chun (2011). "Specification of transplantable astroglial subtypes from human pluripotent stem cells". Nature Biotechnology. 29 (6): 528–534. doi:10.1038/nbt.1877. PMC 3111840. PMID 21602806.. Lay summary: Human Astrocytes Cultivated From Stem Cells In Lab Dish by U of Wisconsin Researchers. sciencedebate.com (22 May 2011)
^ Han J, Kesner P, Metna-Laurent M, Duan T, Xu L, Georges F, Koehl M, Abrous DN, Mendizabal-Zubiaga J, Grandes P, Liu Q, Bai G, Wang Q, Xiong L, Ren Q, Marsicano G, Zhang X (2012). "Acute Cannabinoids Impair Working Memory through Astroglial CB1 Receptor Modulation of Hippocampal LTD". Cell. 148 (5): 1039–50. doi:10.1016/j.cell.2012.01.037. PMID 22385967.
^ Gregori, Ninel; Proschel, Christoph; Noble, Mark; Mayer-Proschel, Margot (January 1, 2002). "The Tripotential Glial-Restricted Precursor (GRP) Cell and Glial Development in the Spinal Cord: Generation of Bipotential Oligodendrocyte-Type-2 Astrocyte Progenitor Cells and Dorsal–Ventral Differences in GRP Cell Function". Journal of Neuroscience. 22 (1): 248–56. PMID 11756508.
^ Fulton, B. P.; Burne, J. F.; Raff, M. C. (1992). "Visualization of O-2A progenitor cells in developing and adult rat optic nerve by quisqualate-stimulated cobalt uptake". The Journal of Neuroscience. 12 (12): 4816–33. PMID 1281496.
^ Levison SW, Goldman JE (1993). "Both oligodendrocytes and astrocytes develop from progenitors in the subventricular zone of postnatal rat forebrain". Neuron. 10 (2): 201–12. doi:10.1016/0896-6273(93)90311-E. PMID 8439409.
^ Zerlin M, Levison SW, Goldman JE (1995). "Early patterns of migration, morphogenesis, and intermediate filament expression of subventricular zone cells in the postnatal rat forebrain". J. Neurosci. 15 (11): 7238–49. PMID 7472478.
^ Brawer JR; Stein, Robert; Small, Lorne; Cissé, Soriba; Schipper, Hyman M. (1994). "Composition of Gomori-positive inclusions in astrocytes of the hypothalamic arcuate nucleus". Anatomical Record. 240 (3): 407–415. doi:10.1002/ar.1092400313. PMID 7825737.
^ Young, J. K.; McKenzie, J. C. (2004). "GLUT2 immunoreactivity in Gomori-positive astrocytes of the hypothalamus". Journal of Histochemistry and Cytochemistry. 52 (11): 1519–24. doi:10.1369/jhc.4A6375.2004. PMC 3957823. PMID 15505347.
^ Marty N (2005). "Regulation of glucagon secretion by glucose transporter type 2 (glut2) and astrocyte-dependent glucose sensors". Journal of Clinical Investigation. 115 (12): 3545–3553. doi:10.1172/jci26309. PMC 1297256. PMID 16322792.
^ Choi BH, Lapham LW (1978). "Radial glia in the human fetal cerebrum: a combined Golgi, immunofluorescent and electron microscopic study". Brain Res. 148 (2): 295–311. doi:10.1016/0006-8993(78)90721-7. PMID 77708.
^ Schmechel DE, Rakic P (1979). "A Golgi study of radial glial cells in developing monkey telencephalon: morphogenesis and transformation into astrocytes". Anat. Embryol. 156 (2): 115–52. doi:10.1007/BF00300010. PMID 111580.
^ Misson JP, Edwards MA, Yamamoto M, Caviness VS (November 1988). "Identification of radial glial cells within the developing murine central nervous system: studies based upon a new immunohistochemical marker". Brain Res. Dev. Brain Res. 44 (1): 95–108. doi:10.1016/0165-3806(88)90121-6. PMID 3069243.
^ Voigt T (November 1989). "Development of glial cells in the cerebral wall of ferrets: direct tracing of their transformation from radial glia into astrocytes". J. Comp. Neurol. 289 (1): 74–88. doi:10.1002/cne.902890106. PMID 2808761.
^ Goldman SA, Zukhar A, Barami K, Mikawa T, Niedzwiecki D (August 1996). "Ependymal/subependymal zone cells of postnatal and adult songbird brain generate both neurons and nonneuronal siblings in vitro and in vivo". J. Neurobiol. 30 (4): 505–20. doi:10.1002/(SICI)1097-4695(199608)30:4<505::AID-NEU6>3.0.CO;2-7. PMID 8844514.
^ Cisneros IE, Ghorpade A (October 2014). "Methamphetamine and HIV-1-induced neurotoxicity: role of trace amine associated receptor 1 cAMP signaling in astrocytes". Neuropharmacology. 85: 499–507. doi:10.1016/j.neuropharm.2014.06.011. PMC 4315503. PMID 24950453.Moreover, TAAR1 overexpression significantly decreased EAAT-2 levels and glutamate clearance that were further reduced by METH. Taken together, our data show that METH treatment activated TAAR1 leading to intracellular cAMP in human astrocytes and modulated glutamate clearance abilities. Furthermore, molecular alterations in astrocyte TAAR1 levels correspond to changes in astrocyte EAAT-2 levels and function.
Further reading
White, F.A.; Jung, H. & Miller, R.J. (2007). "Chemokines and the pathophysiology of neuropathic pain". Proceedings of the National Academy of Sciences of the United States of America. 104 (51): 20151–20158. Bibcode:2007PNAS..10420151W. doi:10.1073/pnas.0709250104. PMC 2154400. PMID 18083844.
Milligan, E.D. & Watson, L.R. (2009). "Pathological and protective roles of glia in chronic pain". Neuron-Glia Interactions. 10 (1): 23–36. doi:10.1038/nrn2533. PMC 2752436. PMID 19096368.
Watkins, L.R.; Milligan, E.D. & Maier, S.F. (2001). "Glial activation: a driving force for pathological pain". Trends Neurosci. 24 (8): 450–455. doi:10.1016/S0166-2236(00)01854-3. PMID 11476884.
Freeman, M. R. (2010). "Specification and Morphogenesis of Astrocytes". Science. 330 (6005): 774–8. Bibcode:2010Sci...330..774F. doi:10.1126/science.1190928. PMC 5201129. PMID 21051628.
Verkhratsky, A.; Butt, A.M. (2013). "Numbers: how many glial cells are in the brain?". Glial Physiology and Pathophysiology. John Wiley and Sons. pp. 93–96. ISBN 978-0-470-97853-5.
External links
| Wikimedia Commons has media related to Astrocytes. |
- Cell Centered Database – Astrocyte
UIUC Histology Subject 57
"Astrocytes" at Society for Neuroscience
- The Department of Neuroscience at Wikiversity
NIF Search – Astrocyte via the Neuroscience Information Framework